题目内容
(请给出正确答案)
题目内容
(请给出正确答案)
为使锅垢中难溶于酸的CaSO4转化为易溶于酸的CaCO3,常用Na2CO3处理,反应式为 CaSO4+ CO32 -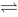 CaCO3+SO42-,此反应的标准平衡常数为 ()
CaCO3+SO42-,此反应的标准平衡常数为 ()
A.KspΘ(CaSO4 ) / KspΘ(CaCO3 )
B.KspΘ(CaCO4 ) / KspΘ(CaSO4 )
C.KspΘ(CaSO4 )KspΘ(CaCO3 )
D.2KspΘ(CaSO4 )/ KspΘ(CaCO3 )
 参考答案
参考答案
 简答题官方参考答案
(由简答题聘请的专业题库老师提供的解答)
简答题官方参考答案
(由简答题聘请的专业题库老师提供的解答)
 网友提供的答案
网友提供的答案
- · 有8位网友选择 C,占比80%
- · 有1位网友选择 B,占比10%
- · 有1位网友选择 A,占比10%



 sp (CaCO3 )
sp (CaCO3 )