题目内容
(请给出正确答案)
题目内容
(请给出正确答案)
One explanation for why graphite powder acts so well as a “solid lubricant” is
A、carbon atoms in graphite are covalently bonded within planar layers but have weaker secondary bonds between layers defect
B、finely-powdered carbon has many unsatisfied bonds at the particle surfaces, which act as a “sea of electrons” to cause lubrication
C、when crushed into a fine powder, graphite establishes a “polar” distribution of charge, leading to Coulombic repulsion between powder particles
D、None of above
 简答题官方参考答案
(由简答题聘请的专业题库老师提供的解答)
简答题官方参考答案
(由简答题聘请的专业题库老师提供的解答)
 抱歉!暂无答案,正在努力更新中……
抱歉!暂无答案,正在努力更新中……

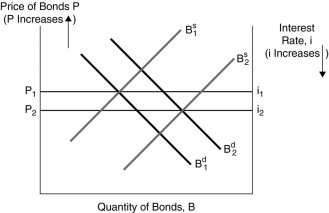 Figure 4.2
Figure 4.2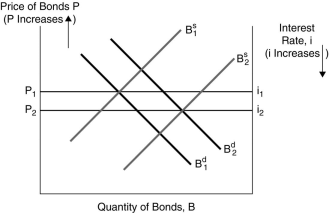 Figure 4.2
Figure 4.2