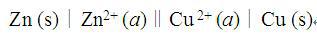题目内容
(请给出正确答案)
题目内容
(请给出正确答案)
用对消法测得原电池 Zn│ZnSO4(a1)‖KCl(a2)│Ag—AgCl(s) 的电动势与温度关系为 E/V=1.0367-5.42×10-4(T/K-298) 则反应 Zn+2AgCl(s)=2Ag+ZnCl2 在 293.2 K 时的焓变为:
A.169.9 kJ/mol
B.-169.9 kJ/mol
C.-231.3 kJ/mol
D.231.3 kJ/mol
 参考答案
参考答案
 简答题官方参考答案
(由简答题聘请的专业题库老师提供的解答)
简答题官方参考答案
(由简答题聘请的专业题库老师提供的解答)
 网友提供的答案
网友提供的答案
- · 有3位网友选择 B,占比33.33%
- · 有3位网友选择 D,占比33.33%
- · 有3位网友选择 A,占比33.33%