 题目内容
(请给出正确答案)
题目内容
(请给出正确答案)
In ideal gas model, all the molecules move in the same directions with a variety of speeds.
 简答题官方参考答案
(由简答题聘请的专业题库老师提供的解答)
简答题官方参考答案
(由简答题聘请的专业题库老师提供的解答)
 抱歉!暂无答案,正在努力更新中……
抱歉!暂无答案,正在努力更新中……
 题目内容
(请给出正确答案)
题目内容
(请给出正确答案)
 简答题官方参考答案
(由简答题聘请的专业题库老师提供的解答)
简答题官方参考答案
(由简答题聘请的专业题库老师提供的解答)
 抱歉!暂无答案,正在努力更新中……
抱歉!暂无答案,正在努力更新中……
 更多“In ideal gas model, all the molecules move in the same directions with a variety of speeds.”相关的问题
更多“In ideal gas model, all the molecules move in the same directions with a variety of speeds.”相关的问题
A、z factor
B、temperature
C、molecular weight
D、pressure
Curves of the Maxwell distribution function for a fixed amount of ideal gas at temperature Ta and Tb are shown in figure. The temperature of ________(a or b) is higher.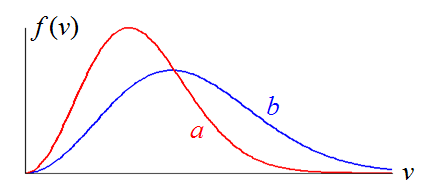
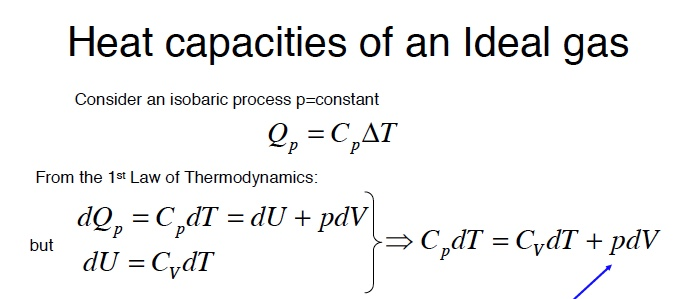 Using the ideal gas law, we can demonstrate Mayer’s relation:
Using the ideal gas law, we can demonstrate Mayer’s relation:
A、Cp – Cv = nR
B、Cp Cv= nR
C、Cp + Cv = nR
D、None the above
A、a junction capacitor, a battery, a small resistor, and the ideal diode
B、a battery, a small resistor, and the ideal diode
C、a battery and the ideal diode
D、the ideal diode
There is a kind of ideal gas in a container with unchangeable volume. When the temperature is at T0 the average speed of the gas molecule is , the average munber of collisions of molecules is
, the average munber of collisions of molecules is and the mean free path is
and the mean free path is . If the temperature inceases to 4T0, what is the average speed
. If the temperature inceases to 4T0, what is the average speed , the average number of collisions
, the average number of collisions and the mean free path
and the mean free path , respectively
, respectively
A、 ,
, ,
,
B、 =2
=2 ,
, =2
=2 ,
, =
=
C、 =2
=2 ,
, =2
=2 ,
, =4
=4
D、 =4
=4 ,
, =2
=2 ,
, =
=

 警告:系统检测到您的账号存在安全风险
警告:系统检测到您的账号存在安全风险
为了保护您的账号安全,请在“简答题”公众号进行验证,点击“官网服务”-“账号验证”后输入验证码“”完成验证,验证成功后方可继续查看答案!
