题目内容
(请给出正确答案)
题目内容
(请给出正确答案)
提问人:网友czq258
发布时间:2022-01-06
[主观题]
称取混合碱试样1.0800 g,溶解后稀释定容至50.00 mL。先移取25.00 mL,加入酚酞作指示剂,用0.1018 m
ol?L-1的HCl标准溶液滴定至终点,消耗HCl 16.82 mL。另取一份25.00 mL,加入甲基橙指示剂滴定至终点,消耗HCl溶液40.68 mL,判断混合碱的组成,并计算试样中各组分的质量分数。 [已知:M(Na2CO3) = 106.0 g/mol;M(NaHCO3) = 84.01 g/mol]
 简答题官方参考答案
(由简答题聘请的专业题库老师提供的解答)
简答题官方参考答案
(由简答题聘请的专业题库老师提供的解答)
 抱歉!暂无答案,正在努力更新中……
抱歉!暂无答案,正在努力更新中……

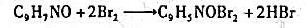
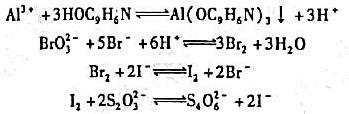
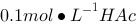
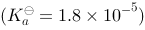 溶液稀释到400 mL,
溶液稀释到400 mL,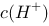 约为原来的 。
约为原来的 。